
李冲
2007.9-2011.5 烟台大学,药学,学士
2012.8-2018.5 美国迈阿密大学医学院,人类遗传学和基因组学,博士
2018.8-2024.8 奥地利科学院分子生物科技研究所,博士后
2024.8-至今 北京脑科学和类脑研究所,研究员
人的大脑拥有约1000亿个神经元,3000多种细胞类型,可形成100兆亿个突触连接,是所有灵长类动物之最。相比其他物种,人脑的发育具有怎样的特殊规律,而这又如何成为了其复杂结构和功能的基础?神经干细胞命运是如何进行精准调控产生并维持细胞谱系的多样性?人脑发育的异常又是如何导致脑疾病,如自闭症和认知障碍?围绕这些脑科学领域的挑战,我们实验室聚焦人脑发育的基本规律和脑疾病的发病机制研究,以人脑类器官(Brain Organoid)这一人源神经生物学模型为基础,结合多功能干细胞工程、CRISPR基因编辑、高通量筛选、单细胞基因组学以及定量生物学等前沿技术,探索人脑特有的运行机制。
核心研究领域:
人脑发育的基本原理:我们将利用多种脑类器官分化技术研究人脑发育所呈现的特有规律及其遗传学基础,探索多种神经元及胶质细胞在人脑结构和功能中作用的特异性。
神经干细胞调控:神经干细胞命运的调控机制对于解析大脑发育和功能至关重要。我们将探索神经干细胞如何分化、增殖、并产生人脑中复杂多样的细胞谱系。
脑疾病机制:我们将从遗传学角度,利用类器官构建脑疾病模型,并在分子、细胞、结构以及神经活动上探索脑疾病的发病机制。
方法学:
I: 人脑类器官
人脑类器官利用干细胞内在全能型,在分化过程中,不仅在组织水平上再现早期人脑结构,同时产生与人脑中高度相似的多种神经干细胞、神经元、神经胶质细胞等细胞类型 (图1)。
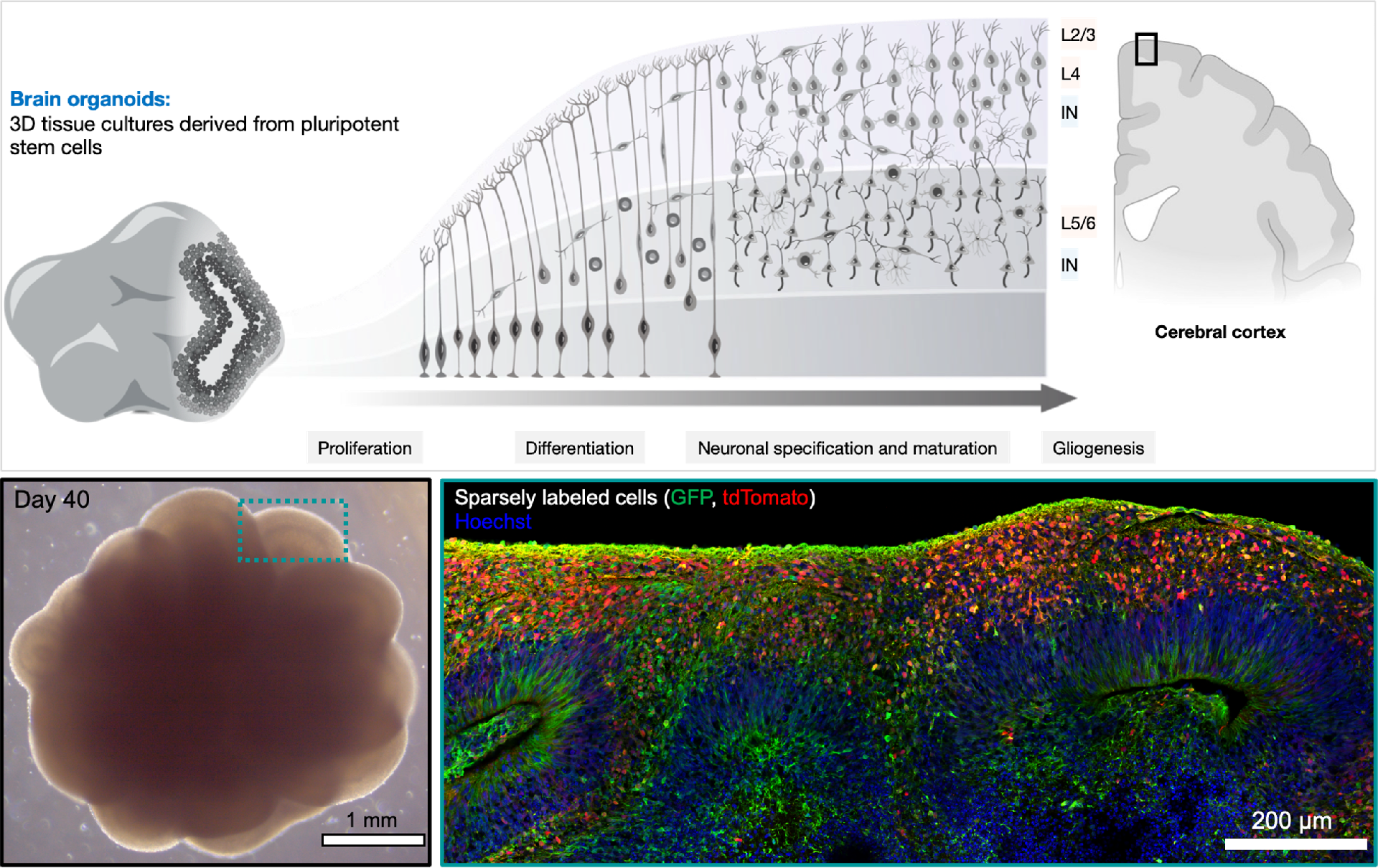
图1
II: 基因编辑、高通量筛选、单细胞组学以及定量生物学
CHOOSE系统将高通量基因编辑和单细胞组学结合在人脑类器官中,使在组织水平上进行基因干扰和功能筛选成为可能(图2)。利用该系统进行的36个高致病性自闭症基因功能筛选研究成功揭示了自闭症中的易感细胞类型、信号通路和基因调控网络 (Li et al., 2023, Nature)。
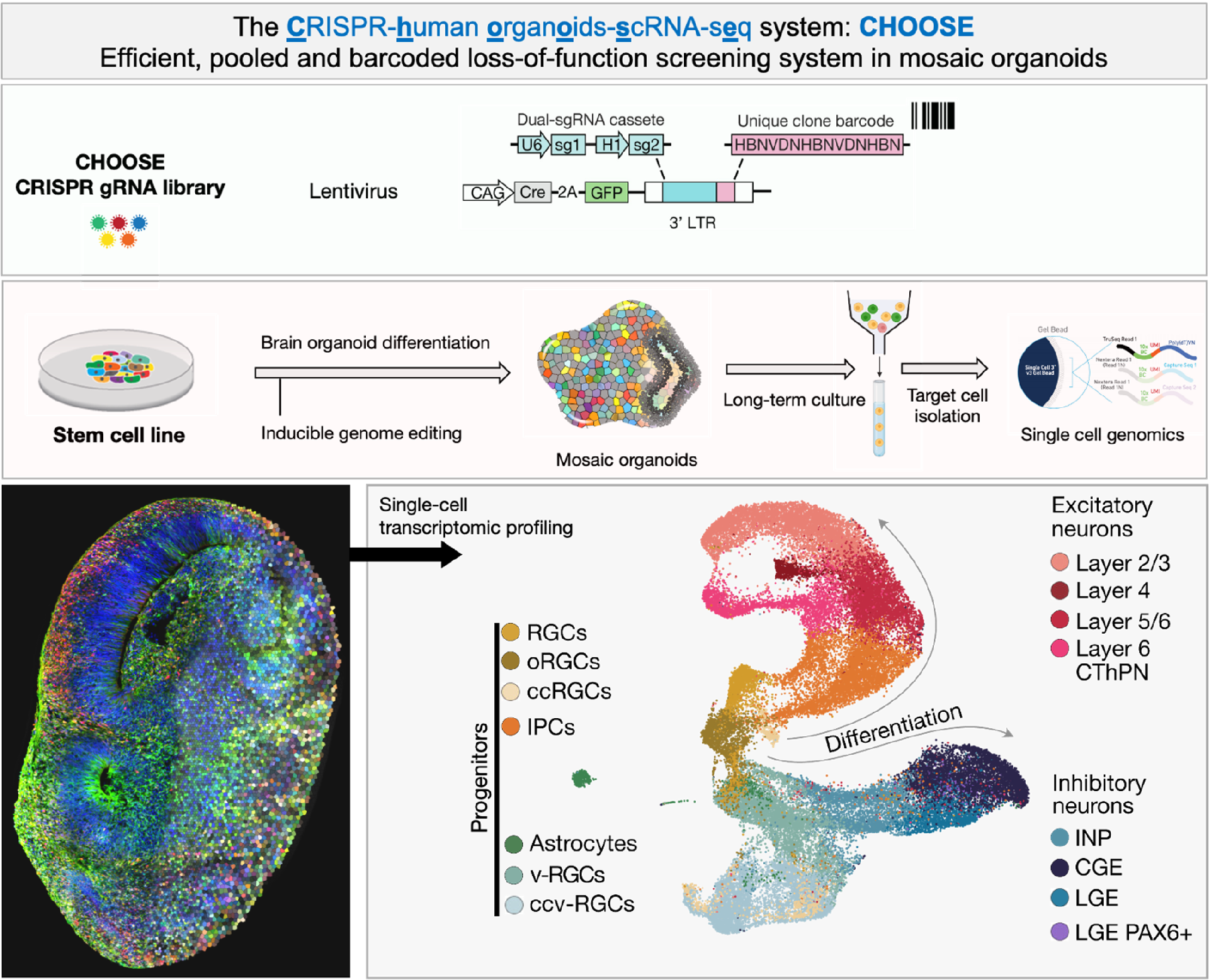
图2
实验室利用创新的模型系统和技术手段,深入探索人脑发育的基本原理和脑疾病的发病机制,致力于为疾病治疗提供理论基础。我们还致力于营造一个充满创新和多样性的科研环境,鼓励实验室成员充分发挥自己的科研兴趣。
欢迎对科研充满热情的博士后、博士生、实习生和科研助理加入我们,一起探索大脑的奥秘。
04/2023 Best Talk Award, Gordon Research Conference, Functional Genomics of Human Brain Development and Disease
11/2017 Best Research Award, Medical Faculty Association, University of Miami
09/2017 American Society for Cell Biology Travel Award
11/2016 Dr. Louis J. Elsas Award in Biochemical Genetics, Dr. John T. Macdonald Foundation
10/2016 Medical Faculty Association Travel Award, The Margaret Whelan Fund
02/2016 Snyder-Robinson Foundation Predoctoral Fellowship, The Snyder-Robinson Foundation
12/2015 Lois Pope LIFE Fellowship Award, Lois Pope LIFE Foundation
1. Li CV#, Knoblich JA#. Advancing autism research: Insights from brain organoid modeling. Curr Opin Neurobiol. 2025 Apr 24;92:103030. doi: 10.1016/j.conb.2025.103030(# co-corresponding)
2. Bosone C*, Castaldi D*, Burkard T R, Guzman S J, Wyatt T, Cheroni C, Caporale N, Bajaj S, Bagley J A, Li C, Sorre B, Villa E, Testa G, Krenn V, and Knoblich J A. (2024) A polarized FGF8 source specifies frontotemporal signatures in spatially oriented cell populations of cortical assembloids Nature Methods 21, 2147–2159
3. Li C*#, Fleck J S*, Martins-Costa C, Burkard T R, Stuempflen M, Vertesy Á, Peer A M, Esk C, Elling U, Kasprian G, Corsini N S, Treutlein B# and Knoblich J A#. (2023) Single-cell brain organoid screening identifies developmental defects in autism. Nature 621, 373–380 (*equal contribution, # co-corresponding)
4. Liu J, Tao X, Zhu Y, Li C, Ruan K, Diaz-Perez Z, Rai P, Wang H, Zhai RG. (2021) NMNAT promotes glioma growth through regulating post-translational modifications of P53 to inhibit apoptosis. Elife. 2021 Dec 17;10:e70046. doi: 10.7554/eLife.70046.
5. Ma X, Zhu Y, Lu J, Xie J, Li C, Shin WS, Qiang J, Liu J, Dou S, Xiao Y, Wang C, Jia C, Long H, Yang J, Fang Y, Jiang L, Zhang Y, Zhang S, Zhai RG, Liu C, Li D. (2020) Nicotinamide mononucleotide adenylyltransferase uses its NAD+ substrate-binding site to chaperone phosphorylated Tau. Elife. Apr 6;9:e51859. doi: 10.7554/eLife.51859.
6. Zhu Y, Li C, Tao X, Brazill JM, Park J, Diaz-Perez Z, Zhai RG (2019) Nmnat restores neuronal integrity by neutralizing mutant Huntingtin aggregate-induced progressive toxicity. PNAS, 116 (38), 19165-19175
7. Park J, Zhu Y, Tao X, Brazill JM, Li C, Wuchty S, Zhai RG (2019) Microrna mir-1002 enhances Nmnat-mediated stress response by modulating alternative splicing. iScience, 19:1048-1064.
8. Li C*, Bademci G*, Subasioglu A, Diaz-Horta O, Zhu Y, Liu J, Mitchell TG, Abad C, Seyhan S, Duman D, Cengiz FB, Tokgoz-Yilmaz S, Blanton SH, Farooq A, Walz K, Zhai RG, Tekin M (2019) Dysfunction of GRAP, encoding the GRB2-related adaptor protein, is linked to sensorineural hearing loss. (*equal contribution), PNAS, 116: 1347-1352
9. Brazill J, Zhu Y, Li C, Zhai RG (2018) Quantitative cell biology of neurodegeneration in Drosophila through unbiased analysis of fluorescently tagged proteins using ImageJ. J. Vis. Exp. (138), e58041, doi:10.3791/58041.
10. Li C, Brazill J, Liu S, Bello C, Zhu Y, Morimoto M, Cascio L, Pauly R, Diaz-Perez Z, Malicdan MCV, Wang H, Boccuto L, Schwartz CE, Gahl WA, Boerkoel CF, Zhai RG (2017) Spermine synthase deficiency causes lysosomal dysfunction and oxidative stress in models of Snyder-Robinson syndrome. Nature Communications, 8(1):1257
11. Gall T, Valkanas E, Bello C, Markello T, Adams C, Bone WP, Brandt AJ, Brazill JM, Carmichael L, Davids M, Davis J, Diaz-Perez Z, Draper D, Elson J, Flynn ED, Godfrey R, Groden C, Hsieh C-K, Fischer R, Golas GA, Guzman J, Huang Y, Kane MS, Lee E, Li C, Links AE, Maduro V, Malicdan MCV, Malik FS, Nehrebecky M, Park J, Pemberton P, Schaffer K, Simeonov D, Sincan M, Smedley D, Valivullah Z, Wahl C, Washington N, Wolfe LA, Xu K, Zhu Y, Gahl WA, Tifft CJ, Toro C, Adams DR, He M, Robinson PN, Haendel MA, Zhai RG and Boerkoel CF (2017) Defining disease, diagnosis, and translational medicine within a homeostatic perturbation paradigm: The NIH Undiagnosed Diseases Program experience. Front. Med., 4:62.
12. Brazill JM*, Li C*, Zhu Y*, Zhai RG (2017) NMNAT: It’s an NAD+ Synthase… It’s a chaperone… It’s a neuroprotector. Curr Opin Genetics Dev., 44:156–162. (*Equal contribution).
13. Ruetenik A, Ocampo A, Ruan K, Zhu Y, Li C, Zhai RG and Barrientos A (2016) Attenuation of polyglutamine-induced toxicity by enhancement of mitochondrial OXPHOS in yeast and fly models of aging. Microbial Cell 3(8):338-351.
14. Ruan K, Zhu Y*, Li C*, Brazill J, and Zhai RG (2015) Alternative splicing of Drosophila Nmnat acts as a switch to enhance neuroprotection under stress. Nature Communications 6:10057 (*Equal contribution)
15. Li C*, Wang T*, Jiang N, Yu PF, Du Y, Ren RT, and Fu FH (2012) Steady and fluctuant methods of inhibition of acetylcholinesterase differentially regulate neurotrophic factors in the hippocampus of juvenile mice. Exp Ther Med, 3(2):269-272. (*Equal contribution)



