
石杨
2012-2018 美国华盛顿圣路易斯大学,神经科学,博士
2007-2011 中国农业大学,生物科学,学士
2022至今 北京脑科学与类脑研究所,研究员
2019.1-2022.6 美国华盛顿圣路易斯大学,博士后
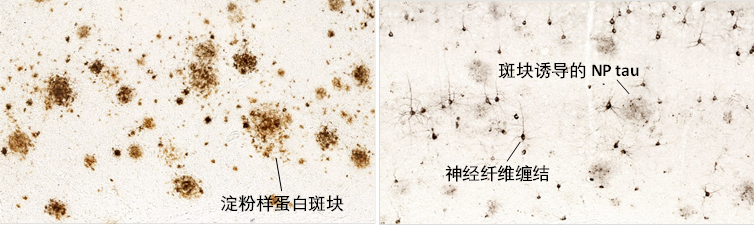
阿尔兹海默症(AD)是当今世界最高发的一种神经退行性疾病, 也是全球最具挑战性的攻坚课题之一。其临床表现为大脑萎缩与认知衰退,在病理上主要表现为大脑内淀粉样蛋白斑块 (Aβ plaque) 在细胞外的堆积,以及tau蛋白在神经细胞内形成的神经纤维缠结(tau tangle),见下图。Aβ plaque 在AD临床症状出现前10-20年开始在大脑内积累,当其在脑内含量达到峰值时,tau病理开始出现,认知损伤与大脑萎缩也随之开始发生。AD的发病机理非常复杂,一个多世纪以来,主流学说一直认为Aβ plaque引发tau病理生成及下游一系列大脑与认知损伤事件,而AD最大的风险因子APOE通过调控Aβ plaque在大脑中的堆积而影响AD患病风险与疾病进程。然而,针对Aβ设计的药物临床试验一直未取得理想结果。
近年来,越来越多的证据表明,免疫系统在很大程度上参与调控AD的发生发展。我们之前在tau蛋白病的小鼠中发现,大脑中存在的免疫细胞-小胶质细胞,是推动tau病理生成和神经退行的真正幕后黑手 (Shi et al, JEM,2019)。当小胶质细胞不存在时,tau病理几乎完全停止进展,且神经退行几乎完全被阻止。而APOE本身具有免疫调节功能,其可通过调控小胶质细胞的激活直接调控tau的病理生成与tau介导的神经退行,这一过程独立于APOE对Aβ plaque的调控 (Shi et al, Nature, 2017)。
除了小胶质细胞在大脑中直接参与调控神经退行,机体的免疫系统在功能上是一个整体,因而周围免疫系统极有可能亦参与调控AD的发生发展。虽然大脑在传统上被认为是免疫豁免的器官,但早期亦有研究发现在正常人大脑的实体组织中存在周围免疫细胞浸润,且其数量在AD病人大脑中显著增加。此外,AD患者血液与脑脊液中周围免疫细胞的组成和激活状态亦有改变。这些证据均提示周围免疫系统可能参与调控AD。
我们实验室通过多学科交叉手段研究神经-免疫轴在阿尔兹海默症中的作用及其机制,以及基于神经免疫的AD创新治疗策略的研发。实验室将重点关注以下研究方向:
1. APOE的免疫调控机制
2. 周围免疫系统对AD病理与神经退行的影响及其作用机制
3. 靶向小胶质细胞及周围免疫系统的治疗方案
4. 神经免疫互作
无论你具有何种学术背景,如果你对阿尔兹海默症和神经免疫研究感兴趣,或者有志于为治疗与预防阿尔兹海默症贡献自己的力量,请联系shiyang@cibr.ac.cn,我们是一个多学科交叉融合的实验室,欢迎来自各领域的你与我们一起头脑风暴!

2021 美国 Rainwater 基金会tau研究领导力奖 (Rainwater Foundation Tau Leadership Award)
2020 美国神经化学学会Marian Kies 奖 (Marian Kies Award of American Society for Neurochemistry)
2019 华盛顿圣路易斯大学神经系Tom Thach 奖 (Tom Thach Award)
2019 第144届美国神经疾病学协会会议旅行奖 (The 144th Annual Meeting of the American Neurological Association Travel Award)
2018 华盛顿圣路易斯大学生物医学研究奖 (David M. Kipnis Award in Biomedical Sciences)
2017 美国 Rainwater 基金会Tau研究协会奖学金 (Rainwater Foundation Tau consortium fellowship)
2016 华盛顿圣路易斯大学神经系Tom Thach 奖 (Tom Thach Award)
1. TCW, J., Qian, L., Pipalia, N., Chao, M., Liang, S., Shi, Y., Jain, B. R., Bertelsen, S. E., Kapoor, M., Marcora, E., Sikora, E., Andrews, E. J., Martini, A. C., Karch, C. M., Head, E., Holtzman, D. M., Zhang, B., Wang, M., Maxfield, F. R., Poon, W. W., Goate, A. M. Cholesterol and matrisome pathways dysregulated in APOE ε4 human microglia and astrocytes. Cell, Accepted, (2022)
2. Shi, Y., Andhey, P.S., Ising, C., Wang K., Snipes, L.L., Boyer, K., Lawson. S., Yamada, K., Qin, W., Manis, M., Remolina J., Benitez, B.A., Schmidt, R.E., Artyomov, M., Ulrich, J., Holtzman, D.M. Overexpressing low-density lipoprotein receptor reduces tau-associated neurodegeneration in relation to apoE-linked mechanisms. Neuron (2021).
Neuron Preview: Mendiola, A. S., Tognatta, R., Yan, Z., Akassoglou, K. ApoE and immunity in Alzheimer’s disease and related tauopathies: Low-density lipoprotein receptor to the rescue (2021).
3. Wang, C., Xiong, M., Gratuze, M., Bao, X., Shi, Y., Andhey, P.S., Manis, M., Schroeder, C., Yin, Z., Madore, C., Butovsky, O., Artyomov, M., Ulrich, J., Holtzman, D.M. Selective removal of astrocytic APOE4 strongly protects against tau-mediated neurodegeneration and decreases synaptic phagocytosis by microglia. Neuron (2021)
4. Litvinchuk, A., Huynh, T., Shi, Y., Jackson, R., Finn, M.B., Manis, M., Francis, C.M., Tran, A., Sullivan, P/M., Ulrich, J., Hyman, B.T., Cole, T., Holtzman, D.M. ApoE4 reduction with ASOs decreases neurodegeneration in a tauopathy model. Annals of Neurology (2021)
5. Wong, M., Lewis, M., Doherty, J. J., Shi, Y, Sullivan, P. M., Covey, D. F., Petsko1, G. A., Holtzman, D. M., Paul, S. M., Luo, W. 25-Hydroxycholesterol amplifies microglial IL-1β production in an apoE isoform-dependent manner. Journal of Neuroinflammation. 17(1):192 (2020)
6. Shi, Y., Manis, M., Long, J., Wang, K., Remolina, J., Hoyle, R., Holtzman, D. M. Microglia drive APOE-dependent neurodegeneration in a tauopathy mouse model. Journal of Experimental Medicine 216 (11): 2546-2561. (2019).
Journal of Experimental Medicine Insights: Kemal, S., Vassar, R. Death by microglia. (2019)
7. Shi, Y & Holtzman, D. M. Interplay between innate immunity and Alzheimer’s disease: APOE and TREM2 in the spotlight. Nature Reviews Immunology 18:759-772, (2018)
8. Shi, Y., Yamada, K., Liddelow, S. A., Smith, S. T., Zhao, L., Luo, W., Tsai, R. M., Spina, S., Grinberg, L. T., Rojas, J. C., Gallardo, G., Wang, K., Roh, J., Robinson, G., Finn, M. B., Jiang H., Sullivan, P. M., Baufeld, C., Wood, M. W., Sutphen, C., McCue, L., Xiong, C., Del-Aguila, J. L., Morris, J. C., Cruchaga, C., Fagan, A. M., Miller, B. L., Boxer, A. L., Seeley, W. W., Butovsky, O., Barres, B. A., Paul, S. M., Holtzman, D. M. ApoE4 markedly exacerbates tau-mediated neurodegeneration in a mouse model of tauopathy. Nature 549, 523-527, (2017).
Science News: A new study is changing how scientists think about Alzheimer's disease (2017). https://www.science.org/content/article/new-study-changing-how-scientists-think-about-alzheimer-s-disease
9. Wang, Y., Ulland, T., Ulrich, J., Song, E., Tzaferis, J., Hole, J., Yuan, P., Mahan, T., Shi, Y., Gilfillan, S., Cella, M., Grutzendler, J., DeMattos, R., Cirrito, J., Holtzman, D., Colonna, M. TREM2-mediated early microglial response limits diffusion and toxicity of amyloid plaques. Journal of Experimental Medicine 213, 667-675, (2016).
10. Jia, S., Liu, Y., Shi, Y., Ma, Y., Hu, Y., Wang M., Li, X. Elevation of Brain Magnesium Potentiates Neural Stem Cell Proliferation in the Hippocampus of Young and Aged Mice. Journal of Cellular Physiology 231, 1903-1912, (2016).
11. Achariyar, T., Li, B., Peng, W., Verghese, P., Shi, Y., McConnell, E., Benraiss, A., Kasper, T., Song, W., Takano, T., Holtzman, D., Nedergaard, M., Deane, R. Glymphatic distribution of CSF-derived apoE into brain is isoform specific and suppressed during sleep deprivation. Molecular Neurodegeneration 11, 74 (2016)
12. Frey, E., Valakh, V., Karney-Grobe, S., Shi, Y., Milbrandt, J., DiAntonio, A. An in vitro assay to study induction of the regenerative state in sensory neurons. Experimental Neurology 263, 350-363, (2015)



