PNAS | 新技术-新发现-新想法:李毓龙、罗敏敏团队揭示神经调质腺苷的释放及调控机制
腺苷(Adenosine, Ado)是一种重要的信号分子,在睡眠、学习记忆、心血管功能及免疫反应等过程中发挥着重要作用。异常的腺苷信号往往伴随着疼痛、癫痫、中风及神经退行性疾病1。早在1929年,Alan Drury和Albert Szent-Györgyi(1937年诺奖获得者)发现,把腺苷类化合物注射到豚鼠中,1-2分钟后豚鼠变得“呼吸急促、无精打采,倾向于安静地躺着、偶尔睡着”,这提示腺苷可能具有调控神经系统的功能2。1974年,John Phillis等人发现,胞外的腺苷能够显著地抑制神经元发放动作电位3;咖啡因、茶碱均可阻断腺苷信号通路,从而起到提神醒脑的功能。尽管科学家对胞外腺苷的功能研究已经非常深入,然而对于腺苷释放如何被调控这一问题,始终存在争议。
2023年3月30日,北京大学李毓龙团队联合北京脑科学与类脑研究中心罗敏敏团队在PNAS杂志在线发表了题为“Neuronal activity-induced, equilibrative nucleoside transporter-dependent, somatodendritic adenosine release revealed by a GRAB sensor”的研究论文。在这项研究中,作者利用新型GRAB荧光探针,详细地研究了神经元活动引发腺苷释放的细胞及分子机制,并揭示了一种非经典的神经调质释放模式。

胞外腺苷来源的争议可以归结为以下几个方面:(1)在细胞水平上,腺苷是来源于神经元还是相邻的胶质细胞?(2)腺苷释放是否具有亚细胞结构的特异性,从突触前还是突触后释放?(3)在分子水平上,腺苷是通过胞外的三磷酸腺苷(ATP)的降解产生;还是通过核苷转运蛋白或突触囊泡直接被释放?
研究胞外腺苷的来源、释放及调控机制的难点在于缺乏可视化检测腺苷的方法。2020年,李毓龙团队和徐敏团队联合报道了一种基于G蛋白偶联受体激活原理(GRAB)的胞外腺苷荧光探针GRAB-Ado1.04,Ado1.0对胞外腺苷的亲和力是~60 nM。在本次报道的研究中,为了进一步扩展探针的检测范围,作者首先通过筛选得到了一个~3.2μM的低亲和力版本Ado1.0m,该探针表现出更大的响应幅度(大于300%)和对腺苷的高特异性。
利用新版Ado1.0m探针,作者成功地在海马急性脑片CA1位置检测到电刺激引发的胞外腺苷水平的上升。有趣的是,作者发现阻断CA3到CA1的谷氨酸突触传递只能部分阻断胞外腺苷水平的上升。那么胞外的腺苷是来源于CA1局部的胞体树突(突触后)还是CA3的轴突投射(突触前)?结合光遗传学工具和胞外腺苷成像,作者发现在阻断谷氨酸突触传递的基础上同时激活CA3轴突,并不能引发腺苷释放;而当激活CA1胞体树突时则发现,胞外腺苷水平表现出刺激时长依赖的增加(图1)。以上结果表明,在亚细胞水平上,胞外腺苷主要来源于CA1的胞体树突(突触后)。
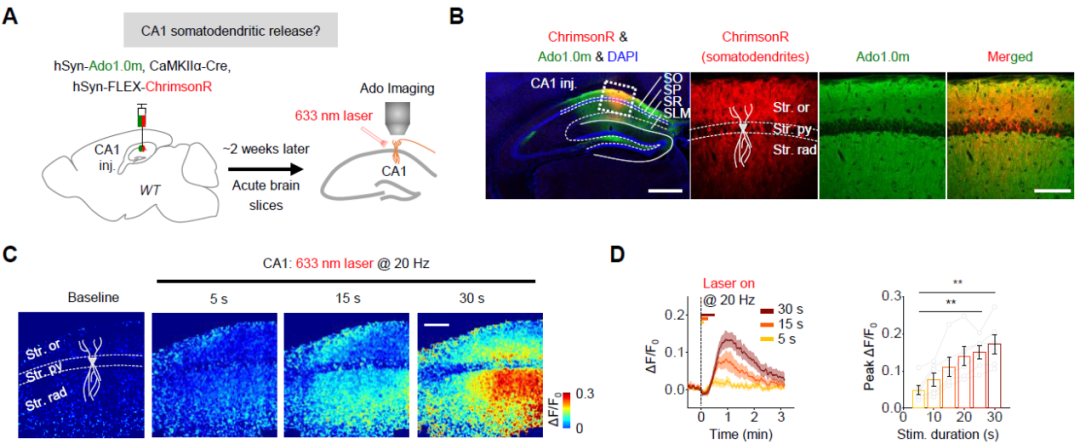
图1:激活CA1胞体树突引发胞外腺苷水平的上升
随后,作者使用培养的海马神经元体系研究胞外腺苷来源的分子机制。结合GRAB腺苷探针、谷氨酸探针及药理学、遗传学等手段,作者发现:在细胞水平上,通过电场刺激能够引发腺苷的释放,而当通过河豚毒素、氯化镉分别阻断表达在神经元上的电压敏感的钠离子和钙离子通道时,腺苷的释放也被阻断,表明胞外的腺苷来源于神经元;当激活星形胶质细胞时,则没有检测到胞外腺苷水平的上升。进一步的,作者还发现胞外腺苷水平的上升主要依赖于的L型钙离子通道,胞内钙库的激活也能引发腺苷的释放。
分子水平上,破伤风神经毒素能够有效地阻断突触囊泡释放的谷氨酸,却无法阻断腺苷的释放,表明腺苷的释放不依赖于SNARE蛋白介导的胞吐过程;阻断CD39或者敲除CD73(胞外ATP降解成腺苷的限速酶)对胞外腺苷的释放没有明显的影响,表明CD39-CD73介导的胞外ATP降解途径对电刺激引发的腺苷释放贡献不大;有意思的是,通过药理学手段阻断或者遗传学手段敲除细胞膜表达的平衡核苷转运蛋白(ENTs),大部分腺苷的释放被阻断,表明在神经元被激活时,胞外腺苷水平的上升主要依赖于ENT转运蛋白(图2)。
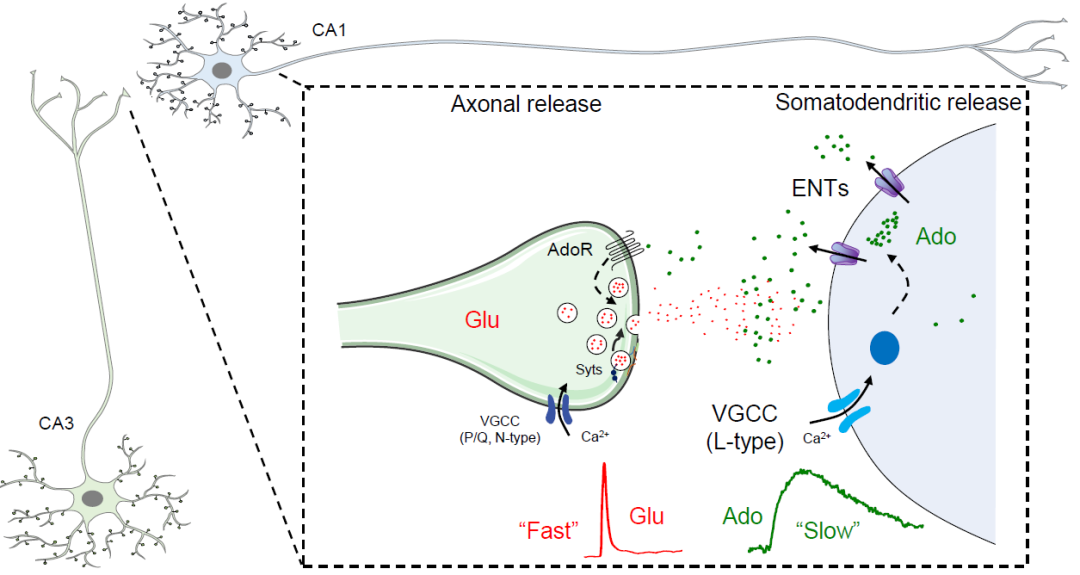
图2:神经元活动引发腺苷释放的模式图
综上,作者发现在海马中神经元活动诱导的胞外腺苷升高主要是由神经元释放的,并取决于ENTs,而不是通过传统的囊泡释放机制;与经典神经递质相比,胞外腺苷积累缓慢(约40秒)并且需要L-型钙离子通道。重要的是,作者发现腺苷主要由突触后直接释放,而不是突触前;基于在突触前膜有丰富的腺苷受体表达,表明突触后释放的腺苷是一个潜在的逆行调控分子(图2)。由于腺苷信号传递及其代谢参与多种生理(睡眠-觉醒调控等)及病理过程(癫痫、中风、疼痛、帕金森病,乃至肿瘤发生等),这项基础研究为理解腺苷的作用机理提供了重要参考,也为腺苷相关疾病的治疗提供了潜在的药物靶点。
相关信息
武照伐博士、崔玉婷博士和王欢博士为本文的共同第一作者,李毓龙教授和罗敏敏教授为共同通讯作者,吴昊、万逸博士、李柏翰、王蕾、潘孙磊、彭婉玲博士、董傲博士、袁正巍博士和井淼博士等对前期探针的开发和刻画做出了重要贡献。论文的合作者还包括中国科学院脑科学与智能技术卓越创新中心的徐敏研究员,并得到了北京大学膜生物学国家重点实验室、北大-清华生命科学联合中心、北京大学IDG麦戈文脑科学研究所、国家自然科学基金、北京市科委、生命医学峰基金、新基石研究员项目、科技创新2030-“脑科学与类脑研究”青年科学家项目、勃林格殷格翰博士后基金和北京市科协“青年人才托举工程”等机构、经费和项目的大力支持。
更多李毓龙实验室工作详见:
罗敏敏实验室工作详见:
https://www.cibr.ac.cn/science/team/detail/36。
此外,李毓龙实验室和罗敏敏实验室诚聘不同学科背景的副研究员、博士后以及科研助理,待遇从优,欢迎对脑科学感兴趣的有志青年加入。
原文地址
https://www.pnas.org/doi/10.1073/pnas.2212387120
参考文献
1. Chen, J. F., Eltzschig, H. K. & Fredholm, B. B. Adenosine receptors as drug targets--what are the challenges? Nat Rev Drug Discov 12, 265-286, doi:10.1038/nrd3955 (2013).
2. Drury, A. N. & Szent-Gyorgyi, A. The physiological activity of adenine compounds with especial reference to their action upon the mammalian heart. J Physiol 68, 213-237, doi:10.1113/jphysiol.1929.sp002608 (1929).
3. Phillis, J. W., Kostopoulos, G. K. & Limacher, J. J. Depression of Corticospinal Cells by Various Purines and Pyrimidines. Canadian Journal of Physiology and Pharmacology 52, 1226-1229, doi:10.1139/y74-162 (1974).
4. Peng, W. et al. Regulation of sleep homeostasis mediator adenosine by basal forebrain glutamatergic neurons. Science 369, doi:10.1126/science.abb0556 (2020).
中外专家点评
陈江帆,
MD/PhD(温州医科大学全职教授、波士顿大学医学院兼职教授)
(译文)在过去的十年里,以“腺苷受体”作为研究主题已有约4500篇科学出版物(与其它GPCR如5-羟色胺的4000篇相当),腺苷信号的研究稳步成熟。腺苷信号控制多种生理过程,包括睡眠、认知、运动和免疫活动,以及抗癫痫和对缺血和神经退行性疾病的神经保护等病理过程。腺苷最为重要的功能是在高频刺激时通过A1受体介导的突触前抑制和主神经元的突触后超极化来抑制神经递质的释放,从而抑制突触传递(特别是兴奋性突触传递)。由于缺乏一种适合检测和可视化细胞分辨率足够的细胞外腺苷的方法,加上大脑腺苷系统在神经调控和稳态调节中的复杂性,使得难以确定在神经元刺激应答中腺苷释放的特定细胞区域和分子途径。
两年前,武博士、李博士及其同事通过GRABAdo(Ado1.0)分子探针的开发取得了重大突破,并在本研究中再次开发了Ado1.0m探针,以便在更广泛的范围内(nM-μM)和亚细胞分辨率下检测腺苷。他们结合新的分子探针和其它尖端方法,包括谷氨酸释放的分子成像、光遗传学和化学遗传学、药理学方法和CRISPR-Cas9策略,提供了迄今为止最有说服力的证据,证明腺苷作为逆行信号的独特作用,作为海马中的神经调质从突触后膜缓慢释放。具体来说,他们修改后的腺苷释放模型包括在细胞和分子水平上确定了多个关键分子靶点,包括神经元细胞,而不是星形胶质细胞,作为腺苷的主要来源;CA1的胞体树突作为释放位点(而不是CA3轴突末梢);L型电压门控钙通道和钙离子通量作为活动依赖性腺苷释放的关键分子级联反应;以及ENT介导跨膜转运的腺苷通过ENT1和ENT2两种途径的关键分子通路,在约40秒内实现缓慢腺苷的释放。对腺苷释放的这些不同特征的分子解析推进了对腺苷系统的理解,这个系统具有神经调质和稳态调控的双重功能,并通过逆行性腺苷信号模型紧密地联系在一起。
此外,他们系统地开发的新型检测腺苷和ATP的工具以及通过CRISPR-Cas9技术操控ENT转运蛋白也是该领域值得关注的贡献。
(原文)There have been ≈4500 scientific publications focused on “adenosine receptors” as a topic over the last decade, which is comparable to other GPCRs such as serotonin/5HT, which has been the subject of ≈4000 publications. This indicates that research on adenosine signaling has been growing steadily and developing over time, reaching a level of maturity. Adenosine signaling controls a wide range of physiological processes, including sleep, cognition, motor function, immune activities, and pathophysiological processes such as anti-convulsant effects and neuroprotection against ischemia and neurodegeneration. The most well-established role of adenosine is its ability to depress synaptic transmission, particularly at excitatory synapses, through the release of adenosine triggered by high-frequency neural stimulation, which is achieved through presynaptic inhibition of neurotransmitter release via A1 receptor-mediated mechanisms and postsynaptic hyperpolarization of principal neurons. The lack of a suitable method for detecting and visualizing extracellular adenosine with sufficient cellular resolution, along with the complexity of brain adenosine systems involved in neuromodulation and homeostasis regulation, has made it difficult to determine the specific cellular compartments and molecular pathways underlying adenosine release in response to neuronal stimulation.
Two years ago, Drs. Wu and Li, along with their colleagues, made a breakthrough with the development of the GRABAdo molecular probe (Ado1.0), and in this recent study, they improved upon it with the Ado1.0m, which is capable of detecting adenosine within a wider range (nM-3µM) and with greater cellular compartment resolution. By utilizing a novel molecular probe, along with other cutting-edge techniques such as molecular imaging of glutamate release, optogenetics, chemogenetics and pharmacological approaches, and CRISPR-Cas9 strategy, they provided the most compelling evidence to date for the unique role of adenosine as a retrograde signaling molecule, which is slowly released from postsynaptic membranes to serve as a neuromodulator in the hippocampus. Specifically, their revised model of adenosine release involves identifying multiple key molecular targets at the cellular and molecular levels, including neuronal cells, rather than astrocytes, as the main sources of adenosine, somatodendritic compartments of CA1 as the release site (rather than axonal terminals of CA3), L-type VGCCs and calcium influx as the critical molecular cascade for activity-dependent adenosine release, and ENT-mediated cross-membrane transport of adenosine via both ENT1 and ENT2 as the key molecular pathway for slow adenosine release throughout ≈40 s. By molecular dissecting the molecular aspects of adenosine release, a new model of retrograde adenosine signaling has been developed, which enhances our understanding of the dual functions of the adenosine system, as both neuromodulator and homeostasis regulator.
In addition, their noteworthy contributions to the field include the development of advanced detection tools for adenosine and ATP, as well as the utilization of CRISPR-Cas9 for ENT1 development.
丁梅,
中国科学院遗传与发育生物学研究所,研究员
众所周知,对突触强度进行快速、持久和有选择性的调适是神经环路形成的基础,也是神经系统对信息编码进行调整以应对环境刺激变化的基础。腺苷由ATP分解代谢形成,也可由从神经元和胶质细胞直接释放。作为信号分子,腺苷通过作用于细胞膜表面的G蛋白偶联受体(A1R、A2AR、A2BR和A3R),调节突触传递和膜被动特性,最终确保生物能量稳态。然而,生理刺激条件下,胞外腺苷如何产生?通过什么方式释放?从什么细胞释放?一直存在争议。
在2020年,北京大学李毓龙课题组就开发了一种基于GPCR激活的腺苷探针GRAB-Ado1.0,可以有效地测量细胞外腺苷水平的变化。现在,李毓龙课题组进一步改进了腺苷探针,并与北京脑科学与类脑研究中心罗敏敏课题组合作,结合光遗传学和胞外腺苷成像,系统、深入地探索了海马体中神经元活动诱导腺苷释放的详细机制。优化之后的腺苷探针(Ado1.0m)特异性更高,对腺苷的响应幅度更大,可对不同组织的胞外腺苷进行精准检测。由于细胞外腺苷参与包括睡眠-觉醒周期、学习和记忆、心血管功能和免疫反应等在内的诸多生理过程,腺苷信号受损也与多种疾病和病理状况,例如疼痛、偏头痛、癫痫、中风、药物成瘾和神经变性息息相关,该探针的进一步广泛应用将有力促进我们对腺苷作用机理的理解,为腺苷相关疾病的治疗提供线索和药物靶点。
Pablo E Castillo,
MD/PhD (Albert Einstein College of Medicine, Harold and Muriel Block Chair in Neuroscience)
(译文)“照亮”大脑中的腺苷信号传递
腺苷是一种强效的神经调质,通过激活广泛分布于中枢神经系统中的G蛋白偶联腺苷受体来调节大脑功能。腺苷信号参与了睡眠-觉醒周期、学习和记忆,并且其失调已被认为与多种脑部疾病相关,包括癫痫、偏头痛、疼痛、药物滥用和神经退行性疾病。虽然腺苷一般会在神经元活动时被释放,但是其精确的机制和来源仍不清楚。为了解决这些关键问题,需要在大脑组织中以高时空分辨率测量腺苷。
近期,北京大学李毓龙实验室开发了一种改进的基因编码腺苷探针(GRAB-Ado1.0m),通过一系列精美而严密的实验表明激活的神经元是细胞外腺苷的主要来源。在他们的实验条件下,其它潜在来源如星形胶质细胞、突触前的释放和ATP降解并不会有显著贡献。此外,作者的研究结果表明,腺苷从胞体树突部位释放,这一过程通过电压门控钙通道引起的去极化诱导的钙离子内流和胞内钙库释放所诱发。尽管导致腺苷产生的钙离子诱导过程尚不清楚,但作者表明平衡核苷酸转运体介导了神经元活动依赖性腺苷释放,而被动性腺苷外流比神经递质释放更为缓慢。胞体树突部位的腺苷释放可以作为逆行信号通过突触前的腺苷受体来调节神经递质的传播。
总的来说,这项新研究为研究大脑中的腺苷信号提供了一个极佳的工具,这也凸显了开发具有适当亲和力、动态范围和时空分辨率探针以在“原位”检测内源性配体存在并更好地表征其功能的重要性。
(原文)Shining light on adenosinergic signaling in the brain
Adenosine is a potent neuromodulator that regulates brain function by activating G protein-coupled adenosine receptors widely distributed in the central nervous system. Adenosinergic signaling is involved in the sleep-wakefulness cycle, learning and memory, and its dysregulation has been implicated in several brain disorders, including epilepsy, migraine, pain, drug abuse and neurodegenerative diseases. While adenosine release typically occurs upon neuronal activity, the precise mechanism and source(s) remain unclear. To address these crucial questions, measuring adenosine with a high spatiotemporal resolution in brain tissue is required.
The Yulong Li lab (Peking University) recently developed an improved genetically-encoded sensor for adenosine (GRAB-Ado1.0m) to demonstrate in a series of elegant and well-controlled experiments that neuronal activation is the primary source of extracellular adenosine (Wu Z et al, Neuronal activity-induced, equilibrative nucleoside transporter-dependent, somatodendritic adenosine release revealed by a GRAB sensor, PNAS 2023). Other potential sources, such as astrocytes, release from presynaptic boutons, and ATP degradation, do not contribute significantly under their experimental conditions. Moreover, the authors’ results demonstrate that adenosine is released from the somatodendritic compartment by depolarization-induced calcium influx via voltage-gated calcium channels and calcium release from intracellular stores. Although the calcium-induced processes that lead to adenosine production remain unclear, the authors show that equilibrative nucleosidase transporters mediate the activity-dependent release of adenosine from neurons and that passive adenosine efflux is comparatively slower than neurotransmitter release. Adenosine release from the somatodendritic compartment could act as a retrograde signal to regulate neurotransmission via presynaptic adenosine receptors.
Overall, this new study provides an excellent tool for studying adenosinergic signaling in the brain. It also highlights the importance of developing sensors with the proper affinity, dynamic range, and spatiotemporal resolution to detect the presence of endogenous ligands in situ and better characterize their function.



